Electrometer
An electrometer
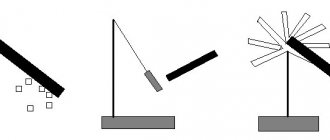
is used to detect and measure electrical charges . The electrometer consists of a metal rod and a pointer that can rotate around a horizontal axis (Fig. 2). The rod with the arrow is fixed in a plexiglass sleeve and placed in a cylindrical metal case, closed with glass covers.
Operating principle of the electrometer
.
Let's touch the positively charged rod to the electrometer rod. We will see that the electrometer needle deviates by a certain angle (see Fig. 2). The rotation of the arrow is explained by the fact that when a charged body comes into contact with the electrometer rod, electrical charges are distributed along the arrow and the rod. Repulsive forces acting between like electric charges on the rod and the pointer cause the pointer to rotate. Let's electrify the ebonite rod again and touch the electrometer rod with it again. Experience shows that with increasing electric charge on the rod, the angle of deviation of the arrow from the vertical position increases. Consequently, by the angle of deflection of the electrometer needle, one can judge the value of the electric charge transferred to the electrometer rod. Rice. 2
Electric charge and its properties.
Electric charge is a physical quantity that characterizes the ability of particles or bodies to enter into electromagnetic interactions. Electric charge is usually denoted by the letters q or Q. In the SI system, electric charge is measured in Coulombs (C). A free charge of 1 C is a gigantic amount of charge, practically not found in nature. Typically, you will have to deal with microcoulombs (1 µC = 10–6 C), nanocoulombs (1 nC = 10–9 C) and picoculombs (1 pC = 10–12 C).
Electric charge has the following properties:.
1. Electric charge is a type of matter.
2. Electric charge does not depend on the movement of the particle and its speed.
3. Charges can be transferred (for example, by direct contact) from one body to another. Unlike body mass, electric charge is not an integral characteristic of a given body. The same body under different conditions can have a different charge.
4. There are two types of electric charges, conventionally called positive and negative.
5. All charges interact with each other. In this case, like charges repel, unlike charges attract. The forces of interaction between charges are central, that is, they lie on a straight line connecting the centers of the charges.
6. There is a minimum possible (modulo) electric charge, called the elementary charge. Its meaning:
e = 1.602177·10–19 C ≈ 1.6·10–19 C.
The electric charge of any body is always a multiple of the elementary charge:
where: N – integer
Please note that it is impossible for a charge equal to 0.5e to exist; 1.7e; 22.7e and so on. Physical quantities that can only take on a discrete (not continuous) series of values are called quantized
Elementary charge e is a quantum (smallest portion) of electric charge.
7. Law of conservation of electric charge. In an isolated system, the algebraic sum of the charges of all bodies remains constant:
The law of conservation of electric charge states that in a closed system of bodies processes of creation or disappearance of charges of only one sign cannot be observed
From the law of conservation of charge, it also follows that if two bodies of the same size and shape, possessing charges q1 and q2 (it does not matter what sign the charges are), are brought into contact and then separated again, then the charge of each of the bodies will become equal:
From a modern point of view, charge carriers are elementary particles. All ordinary bodies are composed of atoms, which include positively charged protons, negatively charged electrons and neutral particles - neutrons. Protons and neutrons are part of atomic nuclei, electrons form the electron shell of atoms. The electric charges of a proton and an electron are exactly the same in magnitude and equal to the elementary (that is, the minimum possible) charge e.
In a neutral atom, the number of protons in the nucleus is equal to the number of electrons in the shell. This number is called the atomic number. An atom of a given substance may lose one or more electrons, or gain an extra electron. In these cases, a neutral atom turns into a positively or negatively charged ion
Please note that positive protons are part of the nucleus of an atom, so their number can only change during nuclear reactions. It is obvious that when bodies are electrified, nuclear reactions do not occur
Therefore, in any electrical phenomena, the number of protons does not change, only the number of electrons changes. Thus, imparting a negative charge to a body means transferring extra electrons to it. And the message of a positive charge, contrary to a common mistake, does not mean the addition of protons, but the subtraction of electrons. Charge can be transferred from one body to another only in portions containing an integer number of electrons.
Sometimes in problems the electric charge is distributed over a certain body. To describe this distribution, the following quantities are introduced:
1. Linear charge density. Used to describe the distribution of charge along the filament:
where: L – thread length. Measured in C/m.
2. Surface charge density. Used to describe the distribution of charge over the surface of a body:
where: S – body surface area. Measured in C/m2.
3. Volume charge density. Used to describe the distribution of charge over the volume of a body:
where: V is the volume of the body. Measured in C/m3.
Note that the mass of the electron is:
me = 9.11∙10–31 kg.
Law of conservation of electric charges
- home
- Directory
- Laws
- Conservation laws
- Law of conservation of electric charges
- Law of conservation of electric charge
- Definitions
- Interaction of electrically charged bodies
- The law of conservation of electric charge in practice
Many physical phenomena observed in nature and the life around us cannot be explained only on the basis of the laws of mechanics, molecular kinetic theory and thermodynamics. These phenomena manifest forces acting between bodies at a distance, and these forces do not depend on the masses of the interacting bodies and, therefore, are not gravitational. These forces are called electromagnetic forces.
Law of conservation of electric charge
Under normal conditions, microscopic bodies are electrically neutral because the positively and negatively charged particles that form atoms are connected to each other by electrical forces and form neutral systems. If the electrical neutrality of a body is violated, then such a body is called an electrified body . To electrify a body, it is necessary that an excess or deficiency of electrons or ions of the same sign be created on it.
Methods of electrification of bodies , which represent the interaction of charged bodies, can be as follows:
- Electrification of bodies upon contact . In this case, during close contact, a small part of the electrons transfers from one substance, in which the connection with the electron is relatively weak, to another substance.
- Electrification of bodies during friction . At the same time, the area of contact between the bodies increases, which leads to increased electrification.
- Influence . The influence is based on the phenomenon of electrostatic induction , that is, the induction of an electric charge in a substance placed in a constant electric field.
- Electrification of bodies under the influence of light . This is based on the photoelectric effect , or photoelectric effect , when, under the influence of light, electrons can fly out of a conductor into the surrounding space, as a result of which the conductor charges.
electrification of a body occurs , then electric charges appear on the bodies, equal in magnitude and opposite in sign.
A negative charge on a body is due to an excess of electrons on the body compared to protons, while a positive charge is due to a lack of electrons.
When a body is electrified, that is, when a negative charge is partially separated from the positive charge associated with it, the law of conservation of electric charge . The law of conservation of charge is valid for a closed system into which charged particles do not enter from the outside and from which they do not leave.
The law of conservation of electric charge is formulated as follows:
In a closed system, the algebraic sum of the charges of all particles remains unchanged:
q1 + q2 + q3 + … + qn = const
where q1, q2, etc. – particle charges.
Definitions
Elementary particles can have electricity. charge, then they are called charged;
Elementary particles interact with each other with forces that depend on the distance between the particles, but exceed many times the forces of mutual gravity (this interaction is called electromagnetic).
Electric charge is a physical quantity that determines the intensity of electromagnetic interactions.
There are 2 signs of electric charges:
- positive
- negative
Particles with like charges repel, and particles with opposite charges attract. A proton has a positive charge, an electron has a negative charge, and a neutron is electrically neutral.
Elementary charge is the minimum charge that cannot be divided.
How can we explain the presence of electromagnetic forces in nature? - All bodies contain charged particles.
In the normal state, bodies are electrically neutral (since the atom is neutral), and electromagnetic forces do not manifest themselves.
A body is charged if it has an excess of charges of any sign:
- negatively charged - if there is an excess of electrons;
- positively charged - if there is a lack of electrons.
Electrification of bodies is one of the ways to obtain charged bodies, for example, by contact).
In this case, both bodies become charged, and the charges are opposite in sign, but equal in magnitude.
Interaction of electrically charged bodies
The interaction of bodies having charges of the same or different signs can be demonstrated in the following experiments. We electrify the ebonite stick by friction on the fur and touch it to a metal sleeve suspended on a silk thread.
Charges of the same sign (negative charges) are distributed on the sleeve and the ebonite stick. By bringing a negatively charged ebonite stick closer to a charged sleeve, you can see that the sleeve will be repelled from the stick (Fig. 1.1).
If you now bring a glass rod rubbed on silk (positively charged) to the charged sleeve, the sleeve will be attracted to it (Fig. 1.2).
The law of conservation of electric charge in practice
Let's take two identical electrometers and charge one of them (Fig. 2.1). Its charge corresponds to 6 scale divisions.
If you connect these electrometers with a glass rod, no changes will occur. This confirms the fact that glass is a dielectric. If you use a metal rod A (Fig. 2.2) to connect the electrometers, holding it by the non-conducting handle B, you will notice that the initial charge will be divided into two equal parts: half of the charge will transfer from the first ball to the second. Now the charge of each electrometer corresponds to 3 scale divisions. Thus, the original charge did not change, it only split into two parts.
If a charge is transferred from a charged body to an uncharged body of the same size, then the charge will be divided in half between these two bodies. But if the second, uncharged body is larger than the first, then more than half of the charge will transfer to the second. The larger the body to which the charge is transferred, the larger part of the charge will be transferred to it.
But the total amount of charge will not change. Thus, it can be argued that the charge is conserved. Those. the law of conservation of electric charge is satisfied.
Electric charges do not exist on their own, but are internal properties of elementary particles - electrons, protons, etc.
In 1914, the American physicist R. Millikan experimentally showed that the electric charge is discrete . The charge of any body is an integer multiple of the elementary electric charge e = 1.6 × 10-19 C.
, the law of charge conservation applies .
qelectron + qpositron = 0.
A positron is an elementary particle with a mass approximately equal to the mass of an electron; The charge of a positron is positive and equal to the charge of an electron.
Based on the law of conservation of electric charge, the electrification of macroscopic bodies is explained.
As you know, all bodies consist of atoms, which include electrons and protons. The number of electrons and protons in an uncharged body is the same. Therefore, such a body does not exhibit an electrical effect on other bodies. If two bodies are in close contact (during rubbing, compression, impact, etc.), then electrons associated with atoms are much weaker than protons and move from one body to another.
The body to which the electrons have transferred will have an excess of them. According to the conservation law, the electric charge of this body will be equal to the algebraic sum of the positive charges of all protons and the charges of all electrons. This charge will be negative and equal in value to the sum of the charges of excess electrons.
A body with an excess of electrons has a negative charge.
A body that has lost electrons will have a positive charge, the modulus of which will be equal to the sum of the charges of electrons lost by the body.
A body that has a positive charge has fewer electrons than protons.
The electric charge does not change when a body moves to another frame of reference.
Laws of conservation Formulas Physics Theory Grade 8 Law Dynamics Mechanics
More interesting things in telegram @calcsbox
Problem solving
Electrostatics is a science that deals with the study and determination of forces that arise when charged particles interact with each other at rest. Using Coulomb's law, it is not necessary to carry out constant measurements for this; it is enough to use the patterns he obtained.
For example, using mathematics and knowledge of the necessary formulas, you can solve the following types of problems:
Determine the force with which electrons carrying a charge of 10-8 coulombs will act on each other if the distance between them is three centimeters. This is a one-step problem, that is, it is solved using one formula: F = k * (q 1 * q 2 / r 2)
It should be noted that the distance is given in centimeters, and it must be substituted according to SI in meters. After substitution and calculations, the answer should be as follows: F = 9 * 109 * (H * m 2 / Cl2) * (10 -8)2 (Cl) / (3 * 10-2)2 (m) = 10-3 N. Find how many times the electrical repulsion between two electrons will be greater than their attractive force
To solve this problem, you will need to take data on the mass of the particle and the magnitude of its charge from the reference book. Then, according to Coulomb’s law, calculate the force of electrical interaction, and according to the law of universal gravitation, the gravitational force and find the ratio of the results obtained. So, F 1 = (k * q 1 * q 2) / r 2 = k * (e)2 / r 2, and F 2 = G * (m 1 * m 2) / r 2 = G * m / r 2. Hence F 1 / F 2 = 9 * 109 * 1.6 * 10-19 / 6.67 * 10-11 * (9.1 * 10-31)2 = 4.23 * 1042. Charged particles are located at each other from each other at a distance of seven millimeters. Calculate the force acting on a 2 nC charge located at a point three millimeters away from a 10 nC charge and four millimeters away from a 16 nC charge. The resultant of the resulting forces can be defined as follows: F = F1 – F2, where: F1 is the interaction with the third of the first charge, and F2 - the second. Thus, the working formula will take the form: F = k * (q 1 * q 3) / r 12 – k * (q1 * q3) / r 22 = k * q 3 * (q 1/ r 12 – q 2/ r 22). After substituting the data in the answer, you should get a force equal to: F = 2 * 10-3 N.
Please note that when substituting source data into formulas, you must adhere to SI
This is important, especially since with the help of dimensions you can check the correctness of the expression used or obtained. And also when solving problems you often have to resort to using a reference book on electrophysics
Other difficulties should not arise when knowing the formulas
And also when solving problems, you often have to resort to using a reference book on electrophysics. No other difficulties should arise when knowing the formulas.
Elementary charge
Avogadro's number and Faraday's constant
If Avogadro's number N
A and Faraday's constant
F
, the amount of elementary electric charge can be calculated using the formula
e = FNA {\displaystyle e={\frac {F}{N_{\mathrm {A} }}}}
(in other words, the charge of one mole of electrons, divided by the number of electrons in a mole is equal to the charge of one electron.)
Compared to other, more accurate methods, this method does not provide high accuracy, but still its accuracy is quite high. Below are the details of this method.
Value of Avogadro's constant N
A was first approximately measured by Johann Joseph Loschmidt, who in 1865 determined on a gas-kinetic basis the size of air molecules, which is equivalent to calculating the number of particles in a given volume of gas[6].
Today, the value of N
A can be determined with very high accuracy using very pure crystals (usually silicon crystals) by measuring the distance between atoms using X-ray diffraction;
or in another way, with an accurate measurement of the crystal density. From here you can find the mass ( m
) of one atom, and since the molar mass (
M
) is known, the number of atoms in a mole can be calculated as follows:
N
A =
M
/
m
.
F value
can be measured directly using Faraday's laws of electrolysis.
Faraday's laws of electrolysis define quantitative relationships based on electrochemical studies published by Michael Faraday in 1834[7]. In an electrolysis experiment, there is a one-to-one correspondence between the number of electrons passing between the anode and cathode, and the number of ions deposited on the electrode plate. By measuring the changes in mass of the anode and cathode, as well as the total charge passing through the electrolyte (which can be measured as the time integral of the electric current), and by taking into account the molar mass of the ions, F
.
Limitations on the accuracy of the method lies in the measurement of F
. The best experimental values have a relative error of 1.6 ppm, which is about thirty times greater than other modern methods for measuring and calculating elemental charge.
Millikan experience
Main article: Millikan experiment
Known experience in measuring electron charge e
.
A small drop of oil in an electric field will move at such a speed that the force of gravity, the Stokes force (derived from the viscosity of air) and the electric force will be compensated. Gravity and Stokes forces can be calculated based on the size and speed of the drop in the absence of an electric field, from which the electric force acting on the drop can be determined. Since the electric force, in turn, is proportional to the product of the electric charge and the known experimentally specified electric field strength, the electric charge of an oil drop can be accurately calculated. In these experiments, the measured charges of various oil droplets turned out to always be integer multiples of one small value, namely e
.
Shot noise
Main article: Shot noise
Any electric current is accompanied by electronic noise from various sources, one of which is shot noise. The existence of shot noise is due to the fact that the current is not continuous, but consists of discrete electrons that alternately enter the electrode. By carefully analyzing the noise of the current, the charge of the electron can be calculated. This method, first proposed by Walter Schottky, can give the value of e
with an accuracy of several percent[8]. However, it was used in Laughlin's first direct observation of quasiparticles implicated in the fractional quantum Hall effect[9].
Josephson effect and von Klitzing constant
Another precise method for measuring the elementary charge is to calculate it from the observation of two effects of quantum mechanics: the Josephson effect, which produces voltage fluctuations in a particular superconducting structure, and the quantum Hall effect, the effect of quantization of Hall resistance or the conductivity of a two-dimensional electron gas in strong magnetic fields and at low temperatures . Josephson constant
KJ = 2 eh , {\displaystyle K_{\mathrm {J} }={\frac {2e}{h}},}
where h
- Planck's constant, can be measured directly using the Josephson effect.
von Klitzing constant
RK = he 2 {\displaystyle R_{\mathrm {K} }={\frac {h}{e^{2}}}}
can be measured directly using the quantum Hall effect.
From these two constants the value of the elementary charge can be calculated:
e = 2 RKKJ. {\displaystyle e={\frac {2}{R_{\mathrm {K} }K_{\mathrm {J} }}}.}
Story
Michael Faraday experimenting in his laboratory
Benjamin Franklin conducts his famous flying kite experiment, in which he proves that lightning is electricity.
Even in ancient times it was known that amber (ancient Greek ἤλεκτρον - electron
), rubbed with wool, attracts light objects.
And already at the end of the 16th century, the English physician William Gilbert called bodies capable of attracting light objects after rubbing as electrified
.
In 1729, Charles Dufay discovered that there are two types of charges. One is formed by rubbing glass on silk, and the other by rubbing resin on wool. Therefore, Du Fay called the charges “glass” and “resin”, respectively. Benjamin Franklin introduced the concept of positive and negative charge.
At the beginning of the 20th century, the American physicist Robert Millikan experimentally showed that the electric charge is discrete
, that is, the charge of any body is an integer multiple of the elementary electric charge.
Quantization of electric charge
Any experimentally observed electric charge is always a multiple of 1 elementary
- this assumption was made by B. Franklin in 1752 and subsequently was repeatedly tested experimentally. The elementary charge was first measured experimentally by Millikan in 1910[3].
The fact that electric charge occurs in nature only in the form of an integer number of elementary charges can be called the quantization of electric charge
. At the same time, in classical electrodynamics the question of the reasons for charge quantization is not discussed, since charge is an external parameter and not a dynamic variable. A satisfactory explanation of why the charge must be quantized has not yet been found, but a number of interesting observations have already been obtained.
- If there is a magnetic monopole in nature, then, according to quantum mechanics, its magnetic charge must be in a certain ratio with the charge of any selected elementary particle
. It automatically follows from this that the mere existence of a magnetic monopole entails charge quantization. However, it was not possible to detect a magnetic monopole in nature. - In modern particle physics, models like the preon are being developed, in which all known fundamental particles would turn out to be simple combinations of new, even more fundamental particles. In this case, the quantization of the charge of the observed particles does not seem surprising, since it arises “by construction.”
- It is also possible that all parameters of the observed particles will be described within the framework of a unified field theory, approaches to which are currently being developed. In such theories, the magnitude of the electrical charge of particles must be calculated from an extremely small number of fundamental parameters, possibly related to the structure of space-time at ultrashort distances. If such a theory is constructed, then what we observe as an elementary electric charge will turn out to be some discrete invariant of space-time (say, topological). This approach is developed, for example, in the model of S. Bilson-Thompson [4], in which the fermions of the Standard Model are interpreted as three ribbons of space-time braided into a braid (braid), and the electric charge (more precisely, a third of it) corresponds to the twisted on 180° tape. However, despite the elegance of such models, specific generally accepted results in this direction have not yet been obtained.
General information
While conducting a series of experiments with amber, William Gilbert noticed that under certain circumstances it was able to attract other objects to itself. In the 16th century, a doctor from England called bodies that had this property electrified. He made the assumption that there is a certain substance in the body that is prone to interaction.
The French scientist Charles Francois Dufay, who did a lot of work on systematizing information related to electricity, came to the conclusion that such a substance is an elementary particle. There are several types of them in each body. As experiments have shown, some were formed by friction of glass on silk, and others by resin. Therefore, the scientist called the particles “glass” and “resin.”
In 1897, physicist Thomson discovered an elementary particle called the electron. Twenty years later, Rutherford suggested the existence of the opposite quantity. After this, the theory was confirmed experimentally, and the particle was named a proton. A scientist from England in 1932 was able to discover a new type of particle, similar in size to the discovered one, but differing in its behavior. He called them neutrons, which translated from Latin means “neither one nor the other.”
After the discovery of current and voltage, scientists discovered interesting effects. The conductor through which electricity flowed caused the magnetic needle to deflect. This phenomenon led to the conclusion that an elementary particle is a carrier of energy, which is called a charge. The concept of positive and negative particles was introduced by Benjamin Franklin.
Thus, it was established that an atom of matter consists of three particles:
- negatively charged electron;
- having a positive charge on a proton;
- neutral particle neutron.
Today, it is understood as a scalar physical quantity that determines the ability of a physical body to be a source of electromagnetic fields. It is the amount of charge that ensures the appearance of electromagnetic interaction. Therefore, it cannot exist without a carrier.
Fractional electric charge
With the discovery of quarks, it became clear that elementary particles can have a fractional electric charge, for example, 1⁄3 and 2⁄3 of an elementary charge. However, such particles exist only in bound states (confinement), so almost all known free particles (and all stable ones) have an electric charge that is a multiple of the elementary one, although scattering by particles with a fractional charge has been observed.
The exception is the t-quark, its lifetime (~1·10−25) is so short that it decays before it has time to undergo hadronization, and therefore is found only in a free form. The t-quark charge according to direct measurements is +2⁄3 e
with a confidence level of about 90%[5].
Repeated searches for long-lived free objects with fractional electric charge, carried out using various methods over a long period of time, did not yield results.
It is worth noting, however, that the electric charge of quasiparticles may also not be a multiple of the whole. In particular, it is quasiparticles with a fractional electric charge that are responsible for the fractional quantum Hall effect.
Protecting radio equipment from electrostatic electricity
Antistatic grounding
Grounding is not a protection against static charges, but it is necessary to limit charges that accumulate on insulating materials and can reach conductive structures of installations.
For static electricity, an object is considered to be grounded if the grounding resistance is on the order of !07 ohms at 60% relative humidity.
To prevent damage to electronic equipment, the following methods are used.
Circuit design
- Use of elements with maximum ESD resistance
- Use of circuit protection against overvoltage in input and output circuits.
Design
- Creation of rational grounding,
- Shielding of nodes and blocks.
- Restricting access to chains and blocks,
- Selection of materials and coatings
Technological
- Elimination of potential differences on materials,
- Use of special containers and transport,
- The use of electrical removal means. static charges from the body of operators,
- Neutralization of charges
- Grounding of objects
Exploitation
- Increasing the surface conductivity of materials by processing materials and creating optimal humidity,
- Neutralization of electricity static charges,
- Antistatic finishing of the room,
- Individual antistatic protection of personnel.
A simple means for removing static electric charge from the body of a human operator.
To remove an electrical charge from the body of a human operator in production, in workshops, and even in a home laboratory, an antistatic bracelet is used.
Its appearance is shown in Fig. 5.
Figure 5
Antistatic wrist strap
The strap of such a bracelet is made of a weakly conductive material, the electrical contact with which is made using a special design that allows you to connect a special wire with a plug to the bracelet. A 1 mOhm resistor should be built into it, which allows the charge to drain from the human body without feeling this process (as happens when a finger directly touches the “ground”).
Attention! Warning for DIYers! Anyone who is trying to make an antistatic bracelet themselves must remember that it cannot be connected with a regular wire to the “ground” or grounding terminal! In an antistatic wrist strap, the connection to ground must be made through a resistance of 1 mOhm! Otherwise, you may lose your hand or die from electric shock!
Links:
- Lecture notes on electrostatics IATE NRNU MEPhI Department of OiSF
- Protection of electronic equipment from the effects of static electricity, Kuchiev L.N., Pozhidaev E.D., Publishing House "Technology", M, 2005
- Circuit protection methods are discussed here.
Collected by A. Sorokin, 2013
Electric charge of an elementary particle
The electric charge of an elementary particle is not a special “mechanism” in the particle that could be removed from it, decomposed into its component parts and reassembled. The presence of an electric charge on an electron and other particles only means the existence of certain interactions between them.
In nature there are particles with charges of opposite signs. The charge of a proton is called positive
, and the electron is
negative
. The positive sign of a charge on a particle does not mean, of course, that it has any special advantages. The introduction of charges of two signs simply expresses the fact that charged particles can both attract and repel. If the charge signs are the same, the particles repel, and if the charge signs are different, they attract.
There is currently no explanation for the reasons for the existence of two types of electric charges. In any case, no fundamental differences are found between positive and negative charges. If the signs of the electric charges of particles changed to the opposite, then the nature of electromagnetic interactions in nature would not change.
Positive and negative charges are very well balanced in the Universe. And if the Universe is finite, then its total electric charge is, in all likelihood, equal to zero.
The most remarkable thing is that the electric charge of all elementary particles is strictly the same in magnitude. There is a minimum charge called elementary
, which all charged elementary particles possess. The charge can be positive, like a proton, or negative, like an electron, but the charge modulus is the same in all cases.
It is impossible to separate part of the charge, for example, from an electron. This is perhaps the most surprising thing. No modern theory can explain why the charges of all particles are the same, and is not able to calculate the value of the minimum electric charge. It is determined experimentally using various experiments.
In the 1960s, after the number of newly discovered elementary particles began to grow alarmingly, it was hypothesized that all strongly interacting particles are composite. More fundamental particles were called quarks. What was striking was that quarks should have a fractional electric charge: 1/3 and 2/3 of the elementary charge. To build protons and neutrons, two types of quarks are enough. And their maximum number, apparently, does not exceed six.
1.1. Electric charge. Coulomb's law
Like the concept of gravitational mass of a body in Newtonian mechanics, the concept of charge in electrodynamics is the primary, basic concept.
Electric charge is a physical quantity that characterizes the property of particles or bodies to enter into electromagnetic force interactions.
Electric charge is usually denoted by the letters or .
The totality of all known experimental facts allows us to draw the following conclusions:
There are two types of electric charges, conventionally called positive and negative.
Charges can be transferred (for example, by direct contact) from one body to another. Unlike body mass, electric charge is not an integral characteristic of a given body. The same body under different conditions can have a different charge.
Like charges repel, unlike charges attract. This also reveals the fundamental difference between electromagnetic forces and gravitational ones. Gravitational forces are always attractive forces.
One of the fundamental laws of nature is the experimentally established law of conservation of electric charge.
In an isolated system, the algebraic sum of the charges of all bodies remains constant:
| . |
The law of conservation of electric charge states that in a closed system of bodies processes of creation or disappearance of charges of only one sign cannot be observed.
From a modern point of view, charge carriers are elementary particles. All ordinary bodies are composed of atoms, which include positively charged protons, negatively charged electrons and neutral particles - neutrons. Protons and neutrons are part of atomic nuclei, electrons form the electron shell of atoms. The electric charges of a proton and an electron are exactly the same in magnitude and equal to the elementary charge.
Electrostatics
Electrostatics is a branch of the study of electricity that studies the interaction of stationary electric charges. Electrostatic (or Coulomb) repulsion occurs between similarly charged bodies, and electrostatic attraction occurs between oppositely charged bodies. The phenomenon of repulsion of like charges underlies the creation of an electroscope - a device for detecting electric charges.
Picture 1
Visual action of Coulomb's law
Electrostatics is based on Coulomb's law. This law describes the interaction of point electric charges.
Coulomb's law looks like:
here ε = 8.85×10-12F/m is the electrical constant.
A simple and clearly observable example of the operation of Coulomb’s law is the transfer of electrostatic charge to a brush made of thin elastic hairs.
Properties of electric charge
There are two types of charge, called positive and negative: charges of the same type repel each other, charges of different types attract, and the repulsive force is equal in magnitude to the attractive force; the number of positive and negative charges in a substance is the same. The total electrical charge of the isolated system is maintained. The charge value can only take discrete values: the minimum particle charge e = 1.60·10-19 C; any charge q is a multiple of the minimum, i.e. q=Ne, where N is an integer; the minimum positive and negative charges are equal in absolute value.
Electric field
The charge changes the properties of the space surrounding it, i.e. he creates something material around himself, through which the interaction between charges occurs. This something is called an electric field. The field is characterized by a strength value that is numerically equal to the force acting on a unit charge:
The direction of the tension vector coincides with the direction of the force acting on the charge.
Value φ
called potential.
The potential (φ) is numerically equal to the potential energy (Wp) that a positive unit charge (q) would have at a given point in the field. The work of transferring charge q from point 1 to point 2 can be written as:
Then, since the potential at infinity is considered equal to zero, we can say that the potential is equal to the work that field forces do on a unit positive charge when it moves away from a given point to infinity.
The unit of potential is the Volt.
1V is the potential at a point, to move to which from infinity a charge of 1C requires 1J of work. The field potential created by a system of charges is equal to the algebraic sum of the potentials created by each of the charges separately:
Electric field strength
Coulomb's law
The possibility of interactions between charges was first confirmed by Coulomb. To do this, he used torsion scales of his own invention. A rocker arm was attached to them, suspended on a silk thread. A micrometer needle was attached to its other end through a clamp. A ball was placed on one pan of the scales, and a counterweight was placed on the other. This entire structure was placed in a vessel from which the air was pumped out. Through a special hole, another ball could be placed into the flask.
As a result of such actions, the physicist observed how, when balls with different charges were placed, the thread twisted. He determined the strength of this interaction by the deflection of the micrometer needle. In addition, with the help of a conductor, Coulomb closed both bodies and observed the distribution of electric charges. Such a device made it possible to measure forces up to 10-11 newtons.
After conducting a series of experiments, the scientist found that the force of interaction is inversely proportional to the square of the distance between the carriers and proportional to the product of their charges. That is, the more energy the carriers have and the closer they are located to each other, the stronger they experience mutual influence. In honor of his work, the energy of a particle began to be measured in coulombs (C).
In mathematical form, the force of interaction is described by the expression: F = k * (q1 * q2) / r2. Where:
- q is the amount of energy that an elementary particle possesses;
- k – coefficient;
- r is the distance between charges.
The coefficient is a constant value, but depends on the choice of measurement system. So, for Gauss it can be calculated using the formula: K = (p * E0) / 4, where E0 is the electrical constant.
Literature
- Burov L.I., Strelchenya V.M. Physics from A to Z: for students, applicants, tutors. – Mn.: Paradox, 2000. – 560 p.
- Myakishev G.Ya. Physics: Electrodynamics. 10-11 grades: textbook. For in-depth study of physics / G.Ya. Myakishev, A.Z. Sinyakov, B.A. Slobodskov. – M.Zh. Bustard, 2005. – 476 p.
- Physics: Textbook. allowance for 10th grade. school and advanced classes studied physicists/ O. F. Kabardin, V. A. Orlov, E. E. Evenchik and others; Ed. A. A. Pinsky. – 2nd ed. – M.: Education, 1995. – 415 p.
- Elementary physics textbook: Study guide. In 3 volumes / Ed. G.S. Landsberg: T. 2. Electricity and magnetism. – M: FIZMATLIT, 2003. – 480 p.
Charge properties
To simplify the description of the field arising around a charged body or even their system, an idealization was introduced - a test point charge. In simple words, this is a quantity with the dimensions of the carrier that can be neglected. In an abstract sense, charge is a generator of continuous symmetry of the physical system being studied. Essentially, it is a substance that “flows” in the physical body.
To study the interaction of charged particles, special instruments are used - an electroscope and an electrometer. The first consists of a metal rod passing through a dielectric plug with two thin metal petals (foil) attached to it. When the body interacts with the rod, the sheets are charged and deviate from each other. The second device uses a pointer that can rotate freely on a rod. By its deviations one judges the magnitude of the electric charge and its influence.
Among the known properties of charged particles, the following can be distinguished:
- There are both negative and positive charges in nature. When interacting, things of the same name are characterized by the appearance of a force of attraction, and those of the same name are characterized by repulsion. The carrier of the smallest negative charge in a physical body is the electron. Its value is q = -1.6*10-19 C, and mass m = 9.1*10-31 kg. The positive one is carried by protons. The value of their charge is similar to that of an electron, but only with the opposite sign, and their mass is greater than m = 1.67*10-27 kg.
- Electric charge is discrete in nature. This means that its value in any case will be a multiple of the electron: q = N qe. In this case, n is always an integer.
- When certain conditions are created, an electric charge can be moved from one body to another.
- In a closed system, the law of conservation of particle energy operates. It has been experimentally established that the appearance of a positively charged particle is accompanied by the disappearance of a negative one. Charges of any sign, if they are equal and interact, can annihilate, that is, neutralize each other.
- A unit of charge is taken to be a value defined as the number of particles passing through the cross-section of a conductor per unit of time.
Experimental determination of elementary electric charge
Avogadro's number and Faraday's constant
If Avogadro's number N
A and Faraday's constant
F
, the magnitude of the elementary electric charge can be calculated using the formula
e=FNA{\displaystyle e={\frac {F}{N_{\mathrm {A} }}}}
(in other words, the charge of one mole of electrons divided by the number of electrons in the mole is equal to the charge of one electron.)
Compared to other, more accurate methods, this method does not provide high accuracy, but still its accuracy is quite high. Below are the details of this method.
Value of Avogadro's constant N
A was first approximately measured by Johann Joseph Loschmidt, who in 1865 determined on a gas-kinetic basis the size of air molecules, which is equivalent to calculating the number of particles in a given volume of gas.
Today, the value of N
A can be determined with very high accuracy using very pure crystals (usually silicon crystals) by measuring the distance between atoms using X-ray diffraction;
or in another way, with an accurate measurement of the crystal density. From here you can find the mass ( m
) of one atom, and since the molar mass (
M
) is known, the number of atoms in a mole can be calculated as follows:
N
A =
M
/
m
.
F value
can be measured directly using Faraday's laws of electrolysis.
Faraday's laws of electrolysis define quantitative relationships based on electrochemical studies published by Michael Faraday in 1834. In an electrolysis experiment, there is a one-to-one correspondence between the number of electrons passing between the anode and cathode, and the number of ions deposited on the electrode plate. By measuring the changes in mass of the anode and cathode, as well as the total charge passing through the electrolyte (which can be measured as the time integral of the electric current), and by taking into account the molar mass of the ions, F
.
Limitations on the accuracy of the method lies in the measurement of F
. The best experimental values have a relative error of 1.6 ppm, which is about thirty times greater than other modern methods for measuring and calculating elemental charge.
Millikan experience
Main article: Millikan experiment
Known experience in measuring electron charge e
.
A small drop of oil in an electric field will move at such a speed that the force of gravity, the Stokes force (derived from the viscosity of air) and the electric force will be compensated. Gravity and Stokes forces can be calculated based on the size and speed of the drop in the absence of an electric field, from which the electric force acting on the drop can be determined. Since the electric force, in turn, is proportional to the product of the electric charge and the known experimentally specified electric field strength, the electric charge of an oil drop can be accurately calculated. In these experiments, the measured charges of various oil droplets turned out to always be integer multiples of one small value, namely e
.
Shot noise
Main article: Shot noise
Any electric current is accompanied by electronic noise from various sources, one of which is shot noise. The existence of shot noise is due to the fact that the current is not continuous, but consists of discrete electrons that alternately enter the electrode. By carefully analyzing the noise of the current, the charge of the electron can be calculated. This method, first proposed by Walter Schottky, can give the value of e
with an accuracy of a few percent. However, it was used in Laughlin's first direct observation of quasiparticles implicated in the fractional quantum Hall effect.
Josephson effect and von Klitzing constant
Another precise method for measuring the elementary charge is to calculate it from the observation of two effects of quantum mechanics: the Josephson effect, which produces voltage fluctuations in a particular superconducting structure, and the quantum Hall effect, the effect of quantization of Hall resistance or the conductivity of a two-dimensional electron gas in strong magnetic fields and at low temperatures . Josephson constant
KJ=2eh,{\displaystyle K_{\mathrm {J} }={\frac {2e}{h}},}
where h
- Planck's constant, can be measured directly using the Josephson effect.
RK=he2{\displaystyle R_{\mathrm {K} }={\frac {h}{e^{2}}}}
can be measured directly using the quantum Hall effect.
From these two constants the magnitude of the elementary charge can be calculated:
e=2RKKJ.{\displaystyle e={\frac {2}{R_{\mathrm {K} }K_{\mathrm {J} }}}.}